This is the seventh national report on organisations funded by the Australian Government to provide health services to Aboriginal and Torres Strait Islander people. In 2014–15: – 203 organisations provided primary health-care services to around 434,600 clients through 5.0 million client contacts and 3.5 million episodes of care; – 221 counsellors provided social and emotional wellbeing services to around 21,100 clients through 100,200 client contacts; – 67 organisations provided substance-use rehabilitation and treatment services to around 25,200 clients through 151,000 episodes of care.
Preference: Administration and Health Services
2
Quality of care achievements of the Prostate Cancer Outcomes Registry–Victoria
Best practice guidelines assist clinicians and health services deliver optimal care to men diagnosed with prostate cancer (CaP),1–3 but little is known about whether optimal approaches to care have been delivered to patients with CaP in Australia. A large United States study that investigated compliance with structural and process of care quality indicators (QIs) highlighted considerable deficits across institutions in care provided to men with localised CaP.4
The Prostate Cancer Outcomes Registry–Victoria (PCOR-Vic), formerly the Victorian Prostate Cancer Clinical Registry (Vic PCR), was established with the aim of improving knowledge of patterns of care and outcomes for men diagnosed with CaP in Victorian hospitals.5 Since its inception in 2009, PCOR-Vic has progressively enlarged its recruitment scope: by 2013 the registry had collected data from 75% of the Victorian target population. To achieve its objectives, a set of QIs was developed after reviewing guidelines and published literature on CaP QIs, and through a consensus process by members of the PCOR-Vic Steering Committee.5 Data on these QIs are reported to contributing clinicians and health services every 6 months after the initial 12-month period following diagnosis.
The registry routinely reports on four outcome measures: patient-reported urinary, bowel and sexual bother, and survival. It also reports on three processes of care indicators. Firstly, it reports to hospitals and clinicians the percentage of men with established low risk disease who have received active treatment during the initial 12-month period after diagnosis (Indicator 1, QI-1). There is now good evidence that active surveillance (AS) is a more appropriate approach than immediate invasive treatment for many men with low risk CaP.6 The Prostate Cancer Research International Active Surveillance (PRIAS) project was initiated in 2006 to describe patterns of care for men on AS and to provide guidance about the AS regimen.7 The Urological Society of Australia and New Zealand has formally endorsed the PRIAS criteria for AS, and has supported the recruitment of patients for PRIAS since 2010.8 Participation in PRIAS is at the discretion of the clinician and patient and thus does not provide a community-based estimate of the uptake of AS.
PCOR-Vic also provides reports about men diagnosed with high risk and locally advanced CaP for whom active treatment was not initiated (Indicator 2, QI-2). The standard approach to treating high risk CaP in patients with an otherwise good life expectancy is prompt initiation of surgery or radiation therapy, the latter with androgen deprivation therapy.2 Delay from the date of biopsy to the date of surgery, particularly for high risk men, may be associated with poorer pathological outcomes.9 There is some evidence that delay in initiating appropriate treatment (based on the risk of disease progression) after the diagnosis of CaP can cause psychological distress.10 More recent evidence indicates that the risk of suicide immediately after diagnosis is 70% higher than for the general population, and that this risk is greatest for men with regionally advanced disease.11
The third process of care indicator routinely measured by the registry relates to risk-adjusted percentages of positive surgical margins (PSMs) following radical prostatectomy (Indicator 3, QI-3). PSM status is important in evaluating quality of care in managing CaP because it is associated with an increased risk of biochemical recurrence after radical prostatectomy12 and is a significant predictor of the need for additional radiotherapy and/or androgen deprivation therapy, even after taking stage of disease into account.13
Patients who are identified as meeting either QI-1 or QI-2 selection criteria are notified to their treating or diagnosing clinicians and hospitals. QI-3 is reported to hospitals and clinicians in the form of risk-adjusted funnel plots if there are more than 20 patients meeting the inclusion criteria. These funnel plots are de-identified with regard to other clinicians, and visually depict variations beyond the mean of two or three standard deviations. Funnel plots were chosen to allow clinicians and health services to compare their performances with those of other providers; to visualise variation in compliance with the indicator; and to stimulate competition in improving outcomes for men with CaP. As with QI-1 and QI-2, hospitals and clinicians are also provided with the names of patients who meet QI-3 criteria (constituting the numerator).
This article reports the overall performance of the Victorian health system over a 5-year period (2009–2013) according to the three processes of care indicators reported by the registry.
Methods
Study population
Men are eligible to be included in PCOR-Vic if they are diagnosed with CaP or treated for CaP at a recruiting hospital after ethics approval at the site; have pathologically confirmed CaP; and are treated by a clinician who has provided consent for patients to be approached and enrolled in the registry. Details of the number of hospitals recruited since 2009 and the average number of patients in each hospital are summarised in Box 1.
Recruitment
The recruitment strategy and data collection methodology of the PCOR-Vic have been previously described.5 In summary, all men identified as having pathologically confirmed CaP notified to the Victorian Cancer Registry and who have not died within 10 months of diagnosis are sent a letter inviting them to participate. We have ethics committee approval to waive patient consent to access management details for men who have died within 10 months; otherwise, an opt-off recruitment strategy is used. Data are collected from hospitals and private consulting suites by trained data collectors. Men are telephoned at 12 and 24 months to confirm that treatment details are accurate and up-to-date, and that they include the most recent prostate-specific antigen (PSA) result.
Statistical analysis
Patients classified as having low risk CaP (ie, if they met the selection criteria of the PRIAS protocol:7 clinical stage T1/T2, PSA ≤ 10 ng/mL, one or two positive biopsy cores, and prostate biopsy Gleason score ≤ 6 based on their most recent biopsy) were included in QI-1. “Most recent biopsy” was defined as the latest biopsy performed during the 12-month period following diagnosis or the initial biopsy at diagnosis if no subsequent biopsy was performed. Men whose initial biopsy or most recent biopsy indicated a higher risk of CaP (ie, no longer met all criteria of the PRIAS protocol) were not included in QI-1. PSA density was omitted in this classification, as the prostate volume (required to calculate PSA density) is not part of the PCOR-Vic minimum dataset.5 Men were deemed to be non-compliant with the PRIAS protocol if they were eligible for PRIAS based on their most recent biopsy and invasive treatment was undertaken within 12 months of diagnosis.
The National Comprehensive Cancer Network (NCCN) definition was used to assess appropriateness of care for high risk and locally advanced disease;1 ie, PSA > 20 ng/mL or Gleason score > 7 or clinical stage ≥ T3a, and not over 80 years of age at diagnosis. Men who met these criteria were included in QI-2, and were deemed to not be receiving appropriate care if active treatment had not been initiated within 12 months of definitive diagnosis of CaP.
Only patients with organ-confined disease (pathological T category 2) were included in the analysis of change in PSM rate over time. Margin status was unequivocal for 2313 patients (98.5%), but in 36 men (1.5%) a definitive status could not be established because the sample was insufficient (four patients), documentation was unclear (seven patients) or the margin status was not stated in the pathology report (25 patients). These cases were not included in further analyses.
Trend analysis was conducted to monitor performance with respect to QI-1, QI-2 and QI-3 for men diagnosed with CaP between 2009 and 2013. Autocorrelation of the month-year data was first assessed by plotting the partial autocorrelation function. We used the autoregressive integrated moving average (ARIMA) model to account for any inherent autocorrelation in the data when analysing the monthly incidence of each indicator. Differences in the annual figures for the indicators across years were also analysed by aggregating data by year and applying the ARIMA model. Data were analysed in Stata 13.0 (StataCorp); P < 0.05 was defined as statistically significant.
Ethics approval
This project received ethics approval from the human research ethics committees of Monash University (reference CF09/0931-2009000436), Cancer Council Victoria (reference 0908) and of each participating health service.
Results
A total of 4708 men were diagnosed with CaP in recruiting hospitals between 1 January 2009 and 31 December 2013 and met at least one of the QI-1, QI-2 or QI-3 inclusion criteria. The PCOR-Vic recorded an opt-off rate of 2.7% over the 5-year period. About 2% of men could not be recruited because their treating doctor had not provided consent for participating in the registry. The demographic and diagnostic details of the men contributing to each indicator are outlined in Box 2.
QI-1: Percentage of men with low risk disease receiving active treatment (non-compliance with PRIAS recommendations)
The proportion of men with low risk disease who underwent active treatment declined between 2009 and 2013. The mean percentage of PRIAS non-compliance had increased non-significantly from 45% in 2009 to 55% (2010) and 50% (2011), but a downward trend was evident in 2012 (40%) and 2013 (34%); this achieved statistical significance in 2013 (P = 0.024).
QI-2: Percentage of men with high risk or locally advanced disease receiving treatment within 12 months of CaP diagnosis
During the 5 years there was a gradual but statistically non-significant increase in the percentage of men with high risk or locally advanced disease receiving treatment within 12 months of diagnosis; 88% of men in these risk groups received immediate active treatment in 2009, rising to 89% in 2010, 91% in 2011 and 2012, and 93% in 2013.
QI-3: Percentage of PSMs for organ-confined pathological T2 disease
The PSM rate for men with organ-confined (pT2) CaP in 2009 was 24%, and rose slightly (by 3.4 percentage points) in 2010. A significant decline was first measured in 2011 (7 percentage point reduction in pT2 prostatectomy PSM; P = 0.031). Further significant improvements were measured for radical prostatectomies performed in 2012 and 2013, when the PSM rate remained steady at 12% (Box 3, Box 4).
Discussion
Given the prevalence of CaP,14 attention to the quality of CaP care has become an important health care issue not only for authorities and policy makers, but also for health service providers and patients. There is broad consensus on the need for regular assessment and monitoring of clinical performance, both for professional development and for quality improvement.15 National data on CaP treatment are limited, and PCOR-Vic was set up as a quality improvement project in 2009 to address this need.5
Our analysis found a downward trend over a 5-year period in the percentage of men with low risk disease who underwent active treatment, an upward trend in the percentage of those with high risk and locally advanced disease who received active treatment within 12 months of diagnosis, and a decline in PSM in men with pathological T2 disease after radical prostatectomy.
The decline in the proportion of men with low risk disease receiving active treatment predated the increased use of transperineal biopsies in Victoria from 2013. AS is a practicable and recommended strategy for reducing overtreatment of patients with low risk CaP.7 The overall level of surveillance of men with low risk CaP in 2012 and 2013 that we found (60%) was higher than that reported by the Michigan Urological Surgery Improvement Collaborative (US) registry which used the D’Amico classification to define low risk CaP (49%).16 Similarly, data from the CaPSURE registry (US) had shown that AS for low risk disease (as defined by the Cancer of the Prostate Risk Assessment [CAPRA] score) had increased sharply from 14.4% in 2009 to 40.4% in 2013.17
We also found that a larger proportion of men in the high risk and locally advanced disease groups were receiving prompt treatment in 2013 (93%) than 5 years previously (88%); the increase, however, was not statistically significant. A number of studies have found that active treatment, such as radical prostatectomy or a combination of external beam radiotherapy and androgen deprivation therapy, was associated with significant benefits in local disease control, reducing the development of metastases, and improving disease-free survival.18,19 Further, mortality among men aged 75 years or more with high risk tumours who receive local therapy is 46% lower than for those treated conservatively, after controlling for age, comorbidities, and risks.20
The change in PSM rate in our study (from 21% in 2009 to 12% in 2012) was similar in proportion to that reported by Cancer Care Ontario (from 37% in 2009 to 21% in 2012).21 Another study that exclusively examined outcomes for patients who had undergone robot-assisted laparoscopic radical prostatectomy procedures reported much lower pT2 prostatectomy margin positivity (4.5% in 2009; 3.0% in 2012)22 than the 12% we found. PCOR-Vic has progressively accrued hospitals, and the number of robot-assisted radical prostatectomy procedures has increased significantly compared with other surgical methods, from 15% of all procedures in 2010 to 60% in 2013. There is evidence that surgical technique, surgeon proficiency and clinical and pathological features of the cancer may affect the risk of a positive margin in patients undergoing radical prostatectomy.23,24
The overall improvements we report across the three QIs are likely to be due to a combination of factors, including an improved knowledge and acceptance of management for low and high risk CaP; technical advances and refinements in surgical technique; and possibly the feedback mechanism provided by the registry, providing hospitals and clinicians with benchmark reports on their performance relative to their peers.
A major strength of our analysis is that PCOR-Vic is a community-based comprehensive quality registry system that captures data about patterns of presentation, management of CaP, treatment procedures, interventions, and patient-reported outcomes related to quality of life, complications, and the effects of CaP treatment on daily living. The data collected by PCOR-Vic are increasingly valuable not only for policy makers and health care providers for guiding best clinical practices and processes and to improve quality of patient care, but also for researchers undertaking relevant investigations of CaP.
Several limitations affect the interpretation of our data. First, the improvement in QIs was detected using PCOR-Vic as a single population, but there may be institutional variations in quality improvement. Second, the process of engagement of hospitals with PCOR-Vic often took a number of months; each hospital started contributing data at different time points that depended on when their individual engagement process was completed. Third, our sample (health services participating in the PCOR-Vic) was restricted to about 75% of the target population, so that generalising our results to the entire target population could be difficult. However, the participating health services are spread broadly across the state and closely reflect the regional/metropolitan and public/private distribution of all CaP care in Victoria.
In conclusion, the community-based PCOR-Vic has identified an overall improvement in the three assessed QIs during its first 5 years of operation. Men in the low risk disease group are less likely to undergo active treatment, showing an increased awareness of adherence to PRIAS recommendations. At the other end of the spectrum, our analysis found that more men with high risk and locally advanced disease are receiving active treatment within 12 months of diagnosis. Finally, the proportion of men with organ-confined (pT2) CaP who had PSM after radical prostatectomy declined across the 5-year period.
Clinical QIs are valuable tools for linking quality registries to improve clinical outcomes. This performance analysis of historical trends in QIs will not only provide important insights into how the PCOR-Vic may have contributed to the outcomes of patients in Victorian hospitals with CaP during its first 5 years of operation, but will also provide the basis for further development of clinical indicators by the binational registry, the PCOR-ANZ,25 and for the implementation of specific evidence-based interventions for optimising CaP care in areas identified as needing improvement.
Box 1 –
Number of hospitals contributing to the Prostate Cancer Outcomes Registry–Victoria, 2009–2013
|
Year of authorisation* |
Number of hospitals (cumulative) |
Average number of consented patients in each hospital |
|||||||||||||
|
|
|||||||||||||||
|
2009 |
9 |
133 |
|||||||||||||
|
2010 |
15 |
91 |
|||||||||||||
|
2011 |
33 |
76 |
|||||||||||||
|
2012 |
33 |
77 |
|||||||||||||
|
2013 |
33 |
67 |
|||||||||||||
|
|
|||||||||||||||
|
* Each hospital has an authorisation date which signifies the earliest date at which a patient’s diagnosis date was considered eligible for participation. |
|||||||||||||||
Box 2 –
Demographic and clinical characteristics of men diagnosed with prostate cancer (CaP) in the Prostate Cancer Outcomes Registry–Victoria, 2009–2013
|
|
|
Low risk disease (Indicator 1) |
High risk and locally advanced disease (Indicator 2) |
Radical prostatectomy (Indicator 3) |
|||||||||||
|
|
|||||||||||||||
|
Total number of contributing men |
1117 |
1840 |
2349 |
||||||||||||
|
Age group |
≤ 55 years |
245 (21.9%) |
109 (5.9%) |
506 (21.5%) |
|||||||||||
|
|
56–65 years |
548 (49.1%) |
543 (29.5%) |
1147 (48.8%) |
|||||||||||
|
|
66–75 years |
307 (27.5%) |
886 (48.2%) |
684 (29.1%) |
|||||||||||
|
|
> 75 years |
17 (1.5%) |
302 (16.4%) |
12 (0.5%) |
|||||||||||
|
PSA at diagnosis |
≤ 4 ng/mL |
289 (25.9%) |
117 (6.4%) |
509 (21.7%) |
|||||||||||
|
|
4.01–10 ng/mL |
828 (74.1%) |
736 (40.0%) |
1584 (67.4%) |
|||||||||||
|
|
10.01–20 ng/mL |
NA |
421 (22.9%) |
193 (8.2%) |
|||||||||||
|
|
≥ 20.1 ng/mL |
NA |
549 (29.8%) |
35 (1.5%) |
|||||||||||
|
|
Unknown |
NA |
17 (0.9%) |
28 (1.2%) |
|||||||||||
|
Gleason score at diagnosis |
≤ 6 |
1117 (100%) |
112 (6.1%) |
856 (36.4%) |
|||||||||||
|
7 (3 + 4) |
NA |
185 (10.1%) |
957 (40.7%) |
||||||||||||
|
|
7 (4 + 3) |
NA |
178 (9.7%) |
315 (13.4%) |
|||||||||||
|
|
8–10 |
NA |
1357 (73.8%) |
209 (8.9%) |
|||||||||||
|
|
Unknown |
NA |
8 (0.4%) |
12 (0.5%) |
|||||||||||
|
Clinical T category |
T1–T2a |
1052 (94.2%) |
643 (34.9%) |
1570 (66.8%) |
|||||||||||
|
|
T2b–T2c |
65 (5.8%) |
370 (20.1%) |
256 (10.9%) |
|||||||||||
|
|
T3a |
NA |
445 (24.2%) |
30 (1.2%) |
|||||||||||
|
|
T3b–T4 |
NA |
82 (5.0%) |
1 (< 0.1%) |
|||||||||||
|
|
Unknown |
NA |
290 (15.8%) |
492 (20.9%) |
|||||||||||
|
NCCN risk group |
Low |
1047 (93.7%) |
NA |
563 (24.0%) |
|||||||||||
|
|
Intermediate |
70 (6.3%) |
NA |
1350 (57.5%) |
|||||||||||
|
|
High |
NA |
1748 (95.0%) |
256 (10.9%) |
|||||||||||
|
|
Locally advanced |
NA |
92 (5.0%) |
1 (< 0.1%) |
|||||||||||
|
|
Metastatic |
NA |
NA |
10 (0.4%) |
|||||||||||
|
|
Unknown |
NA |
NA |
169 (7.2%) |
|||||||||||
|
Diagnostic method |
Transrectal ultrasound (TRUS) |
1102 (98.7%) |
1692 (92.0%) |
2278 (97.0%) |
|||||||||||
|
|
Transperineal template biopsy |
15 (1.3%) |
35 (1.9%) |
19 (0.8%) |
|||||||||||
|
|
Transurethral resection of the prostate (TURP) |
NA |
100 (5.4%) |
42 (1.8%) |
|||||||||||
|
|
Transurethral resection of bladder tumour (TURBT) |
NA |
4 (0.2%) |
NA |
|||||||||||
|
|
Other |
NA |
3 (0.2%) |
4 (0.2%) |
|||||||||||
|
|
Unknown |
NA |
6 (0.3%) |
6 (0.3%) |
|||||||||||
|
Surgical approach |
Open prostatectomy |
NA |
NA |
1013 (43.1%) |
|||||||||||
|
|
Laparoscopic prostatectomy |
NA |
NA |
150 (6.4%) |
|||||||||||
|
|
Robot-assisted laparoscopic prostatectomy |
NA |
NA |
1179 (50.2%) |
|||||||||||
|
|
Other prostatectomy |
NA |
NA |
6 (0.3%) |
|||||||||||
|
|
Unknown type of prostatectomy |
NA |
NA |
1 (< 0.1%) |
|||||||||||
|
Number of men for year of diagnosis (Indicators 1 and 2) or surgery (Indicator 3) |
|||||||||||||||
|
|
2009 |
164 (14.7%) |
240 (13.0%) |
188 (8.0%) |
|||||||||||
|
|
2010 |
181 (16.2%) |
256 (13.9%) |
287 (12.2%) |
|||||||||||
|
|
2011 |
353 (31.6%) |
474 (25.8%) |
672 (28.6%) |
|||||||||||
|
|
2012 |
264 (23.6%) |
467 (25.4%) |
661 (28.1%) |
|||||||||||
|
|
2013 |
155 (13.9%) |
403 (21.9%) |
541 (23.0%) |
|||||||||||
|
|
|||||||||||||||
|
NA = not applicable; NCCN = National Comprehensive Cancer Network; PSA = prostate-specific antigen level. |
|||||||||||||||
Box 3 –
Changes in QI-1, QI-2 and QI-3 between 2009 and 2013, using an autoregressive integrated moving average (ARIMA) model
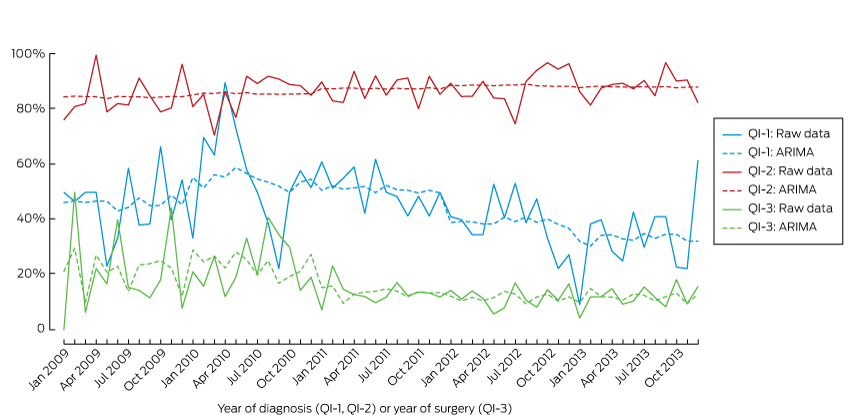
QI-1 = Men with low risk prostate cancer (CaP) who underwent active treatment; QI-2 = men with high risk or and locally advanced CaP who underwent active treatment within 12 months of diagnosis; QI-3 = positive surgical margins in organ-confined (pT2) disease.
Box 4 –
Changes in the three process quality indicators QI-1, QI-2 and QI-3 (baseline: 2009)
|
Year |
QI-1 |
QI-2 |
QI-3 |
||||||||||||
|
% |
Change since 2009 (95% CI) |
P* |
% |
Change since 2009 (95% CI) |
P* |
% |
Change since 2009 (95% CI) |
P* |
|||||||
|
|
|||||||||||||||
|
2009 |
45.1% |
|
|
88.3% |
|
|
20.9% |
|
|
||||||
|
2010 |
55.3% |
10.2 (–1.6 to 17.7) |
0.101 |
89.5% |
1.2 (–4.0 to 6.5) |
0.648 |
24.3% |
3.4 (–1.1 to 5.9) |
0.170 |
||||||
|
2011 |
50.4% |
5.3 (–10.4 to 20.0) |
0.536 |
90.7% |
2.4 (–2.2 to 8.3) |
0.251 |
13.8% |
–7.1 (–14.9 to –0.7) |
0.031 |
||||||
|
2012 |
39.8% |
–5.3 (–20.6 to 6.3) |
0.297 |
90.8% |
2.5 (–0.4 to 8.8) |
0.074 |
12.0% |
–8.9 (–17.2 to –2.2) |
0.012 |
||||||
|
2013 |
34.2% |
–10.9 (–23.4 to –1.7) |
0.024 |
92.8% |
4.5 (–1.7 to 9.1) |
0.181 |
11.7% |
–9.2 (–18.1 to –0.6) |
0.036 |
||||||
|
|
|||||||||||||||
|
QI-1 = Men with low risk prostate cancer (CaP) who underwent active treatment; QI-2 = men with high risk or and locally advanced CaP who underwent active treatment within 12 months of diagnosis; QI-3 = positive surgical margins in organ-confined (pT2) disease. * For change since 2009. |
|||||||||||||||
Forsaking cures for cancer: why are we discarding the tumour biospecimens of most patients?
Discarding patients’ cancer samples because of problems with consent processes forgoes potential advances in cancer research, ignores public intent and squanders unique bioresources
Cancers are highly variable — between different types of cancer, between different patients’ cancers and even between different cancer cells within an individual patient’s cancer.1 A critical challenge facing cancer research and therapy is to understand and overcome the heterogeneity of each patient’s cancer. Resected tumours and other samples donated by patients with cancer provide invaluable bioresources for the study of cancer heterogeneity. While animal models and in vitro studies generate therapeutic hypotheses, only confirmation in human cancers can ratify targets as relevant to pursue into the clinic.
Biobanking processes ensure the collection, annotation, management, appropriate storage and transfer of donated (cancer and healthy) tissue and other samples, which are often those resected at surgery. Revolutionary cancer treatments such as mutation-targeted therapies and checkpoint immunotherapy antibodies could not have been developed without studying biobanked biospecimens. Conversely, prospectively annotated high-quality cancer biospecimens are key to understanding which patients will benefit from existing drugs (drug individualisation) and which patients might forgo otherwise toxic therapies altogether (treatment de-escalation).
The value of biobanks to medical research, research communities and public health has been widely recognised. Biobanks can be viewed as both the primary facilitators of biomedical research efforts and the conduits between cancer patients, as contributors of bioresources, and researchers, as recipients. The rate of successful biobanking is proposed as a key performance indicator,2 but the proportion of Australian patients donating tissue at the time of surgical treatment for cancer is poorly reported and, although largely unknown, appears to be low even in highly motivated and research-focused academic centres. Rather than being systematic, biobanking is often opportunistic, driven by individual clinician–researchers, tissue bank professionals and pathology staff. Biobanking seems to be relatively uncommon in private practice, even though a significant proportion of all cancer surgery occurs outside the public sector.
This picture is at odds with the intent and spirit of patients with cancer, their advocates and the Australian public. Recent studies show that Australians, including both cancer tissue donors (95%) and the general public as potential donors (85%), are willing to donate samples for future unspecified research that is approved by a human research ethics committee (HREC).3 Consent rates as high as 99% have been reported.3 This willingness is likely to be altruistically driven by a sense of duty as a citizen and by trust, which can, if tested, be easily eroded.
Biobanks in Australia have been supported by philanthropic and public donations and by governments.4 Despite widespread recognition of their contributions to research, the future sustainability of cancer biobanks in Australia is, at best, uncertain. While biobanking was initially supported through National Health and Medical Research Council (NHMRC) Enabling Grants, funding support for established biobank entities, including members of the Australasian Biospecimen Network Association – Oncology Group, remains tenuous. While organisations such as the state-based cancer councils and cancer institutes have provided much needed support, infrastructure and encouragement, essential funding remains intermittent.
The barriers to biobanking participation extend beyond funding. Although we are moving to national strategies for biobanking,4 many policies and processes are often reinvented at local and statewide levels. However, issues of patient consent appear to be a nationally important barrier between high rates of public approval and low rates of biospecimen collection. Broad consent for biobanking would allow researchers timely access to tissue for ethically approved research without the need to obtain retrospective consent for specific projects, which is costly and neither helpful nor feasible, particularly in patient populations with highly morbid diseases such as cancer. Asking patients to reconfirm consent for access to donated specimens for new or extended studies could be distressing and intrusive to the patients and their families. Asking individuals to reconsent to biobanking and questioning their decision may create uncertainty and compromise the timely conduct of research.
Given the high levels of willingness to donate and trust in biobanking that have been reported in studies among the Australian general public, we advocate a waiver of consent for biobanking. Although this may be provocative to some,5 consent for biobanking is already embedded in existing routine admission forms of our public hospitals, and promoting awareness of this should be a priority. Consideration should be given to the idea that consent for surgical procedures is sufficient to allow the routine storage of biomaterials (eg, resected tumour tissue and left-over diagnostic samples) for use in ethically and scientifically robust research approved in future by an HREC (ie, conditional donation). Likewise, data linkage to a patient’s medical record and other medical resource usage can be made in a confidential, encrypted and secure manner,6 where results would be reported in aggregate form to protect patients’ identities but could be potentially re-identifiable if clinically relevant incidental findings required reporting to an individual donor.
National regulation of consent processes provided by the NHMRC National statement on ethical conduct in human research (chapter 2.3)7 has for some time offered guidance to researchers and members of HRECs when considering the circumstances in which a waiver for consent can be justified. While recognising that waiving of consent should always be adopted in a measured fashion,8 there is additional scope to waive consent, particularly where the research involves minimal harm to participants.7 To date, reasons for the inconsistent interpretations of the Statement (and hesitancy in its application) by both researchers and HRECs remain unclear and warrant examination.
Applying a routine consent waiver to the hospital admission process could employ a two-step model, wherein (1) collected samples are automatically stored for research, but (2) pending confirmation of patient consent. This may include an opt-out provision that would require authorisation by the patient at some (negotiable) point during his or her admission. This process would also promote greater awareness among patients and health carers of the benefits of biobanking samples by:
-
collecting biospecimens from material that has been taken for diagnostic testing, which would be linked to the patient’s medical record and flagged for future unspecified research approved by an HREC;
-
respecting patient preferences and consideration of choice in the type of consent to be obtained at the time of initial contact; and
-
providing more seamless and fluid consent processes to minimise unnecessary distress to patients and next of kin, as well as to staff who may be in the position of asking difficult questions at difficult times.
Biobanking has also been the subject of sustained scientific and ethico-legal critique, much of which has focused on the means of obtaining consent for long term storage and future use of tissue. Ethical disagreements frequently centre on the tension between individual autonomy and the “common good”. This tension is significant in the context of biobanking research, which often depends on the participation of large numbers of people (including healthy populations), many of whom are unlikely to benefit from the results of the research.
Persistent and emerging issues seek resolution — these include managing an incidentally identified germline heritable mutation, ensuring data linkage security and ensuring that a patient’s genetic makeup does not cause discrimination or economic harm (eg, denial of insurance). International assessments, however, suggest that the general public still overwhelmingly supports the public good of biobanking and is less concerned with privacy and confidentiality.9
In 2003, the Essentially Yours report10 included numerous submissions from key agencies engaged in ethico-legal debates concerning biobanks, highlighting the critical need to deal with the failure of informed consent processes for tissue collections in public tertiary hospitals. Indeed, the Association of Australian Medical Research Institutes (submission G007) acknowledged that discarding samples could be seen as wasteful and that it would be disrespectful to the donors (including deceased donors) if research on sample collections did not take place, or if there was further insistence on gaining consent from relatives.
An economic argument is also pertinent. With over 40 000 surgical procedures for cancer in Australia every year,11 and a cost to cancer researchers of up to hundreds or thousands of dollars per biospecimen (depending on the economic model used),12,13 millions of dollars of invaluable and irreplaceable resources are being squandered in Australia annually. This must be set against the cost of collecting and maintaining biobanked specimens, which is also hard to quantify. Finally, with rapidly increasing cancer drug prices,14 biobanked specimens will drive research to individualise, optimise and de-escalate treatments, which will be essential for improving and economising cancer treatment in an environment of health care expenditure cost containment.
Cancer biobanks are custodians of invaluable, well annotated biospecimen collections. Community engagement has provided empirical evidence about the consent preferences of actual tissue donors, patients with cancer and the general public (as potential donors). Promoting a shift away from the current focus on locally variable, opt-in, physician-driven consent processes is warranted. Acknowledging the shared common good, humanity and responsibility of biobanking could markedly improve our current practices and enable further advancements. Discarding patients’ unique cancer samples forgoes potential advances in cancer research, ignores the public intent, squanders unique resources and is economically wasteful.
Lost in translation: the gap between what we know and what we do about cardiovascular disease
An estimated one million Australians at high risk may not have been receiving recommended preventive treatments
We still seem disappointingly distant from getting it right with disease prevention in Australia. An analysis of data from the 2011–12 Australian National Health Measures Survey reported in this issue of the MJA1 found that most respondents at high risk of cardiovascular disease (CVD) were not receiving the recommended combination of blood pressure-lowering and lipid-lowering medications. Applying these estimates to the general population, almost one million Australians at high risk may not have been receiving these treatments, although their use has long been recommended by the national peak body on the basis of level A evidence.
Perhaps the data are incorrect? While the response rate to the survey among those aged 45–74 years (47%) was lower than desirable, survey respondents would be expected to have better, not worse, treatment rates than non-respondents. It is also likely that self-reporting overestimates treatment levels. Perhaps the data reflect the prevalence of contraindications to recommended treatment? Again, this is unlikely: definite contraindications to lipid-lowering and blood pressure-lowering medications are very rare, and placebo-controlled trials have shown that only a small proportion of subjects taking statins experience symptomatic side effects.2
What is going wrong?
How much of the problem is related to physicians not prescribing medications in the first place, and how much is due to patients not continuing to use them in the long term? For people with an acute coronary syndrome, prescription rates at discharge have climbed steadily in recent years; the main challenge seems to be the steady drop-off in adherence in the months and years following the acute event.3 Both patient- and prescriber-related factors contribute to this drop-off. Patient-related factors include comorbidities, mental health problems, social context, and their perceptions of their illness; even those who have a heart attack underestimate their risk, and misperceptions are associated with the persistence of risk-elevating behaviours, including smoking and non-adherence to medication.4 The cost of drugs may play a bigger role in the Australian context than is recognised.5 Physicians may fail to recognise non-adherence or explain the need to continue therapy. Over-attribution of “side effects” to medication is also a factor,2 compounded in Australia by biased media coverage about statins.6
For people at high primary risk of CVD, we suspect the main problem is under-prescribing of medications that reduce the risk of CVD. A paradigm change is needed, and we have only begun to address this need. The dominant paradigm for many decades has been to prescribe these medications only for people with hypertension or dyslipidaemia; indeed, many drug labels still recognise only these indications. The compelling rationale that use of these drugs should instead be primarily targeted on the basis of high absolute risk of CVD has been plain for many years. The evidence for the benefits of blood pressure-lowering medications for people with non-optimal blood pressure but without hypertension has become even clearer in the past few months.7 Nevertheless, diagnosis and treatment practices for hypertension remain similar to those of several decades ago, when it was erroneously thought there was a threshold of hypertension below which lowering blood pressure conferred no benefit.
Do we need to try something new?
We clearly need to improve our strategies for addressing the evidence–practice gap in CVD risk management. This is part of a wider systemic problem in continuing medical education, our ability to translate large amounts of new research data into practice.
Should we be considering a public health and policy-driven approach? In the United Kingdom, the Quality and Outcomes Framework (QOF) annually rewards practices for their provision of quality care according to evidence-based indicators.8 For example, the indicators for the secondary prevention of coronary heart disease (CHD) include general practices maintaining a register of CHD patients and of the proportions prescribed angiotensin-converting enzyme inhibitors or angiotensin-receptor blockers, aspirin, β-blockers, and statins.8 A registry would enable practices to have annual recall programs for reviewing treatment of CHD patients. In Australia, the National Health and Medical Research Council has recently adopted implementing the absolute risk approach to primary prevention as a priority for research translation.9
Technology-based solutions may also help. Hospital medical records, patient-controlled e-health medical records, and general practice medical records could incorporate red flags for pre-specified indicators of prevention needs not being met. Electronic decision support studies support the potential effectiveness of these type of strategies.10 Other technology-based solutions could include smart pill boxes, pharmacy support programs, telehealth support, and automated text reminders.11 These strategies are all ready to roll now, whereas other innovations, such as polypills, may play a role in the near future.12 There is evidence that supports the efficacy of all these strategies, but none has yet been substantively employed.
CVD is our leading killer and cause of health care costs, but almost one million Australians at high risk may not have been receiving the basic recommended preventive medications. This is the low-hanging fruit in CVD control. A multifaceted approach is needed: patient-centred, evidence-based primary care will be the key.
Futility and utility
The physician should focus on what can be done, not on what cannot
“Futility” is a term that has come into prominence in the medical literature over the past two to three decades. This rise has been led primarily by medical ethicists, and has particularly focused on its place in end-of-life discussion.1 Futility is addressed in this issue of the MJA by White and colleagues,2 who examined its use in a Brisbane hospital by undertaking a survey and interviews with 96 public hospital specialists and trainees from 16 specialties. They found that the term was widely employed, but also that there was some confusion about its meaning and when to invoke it. It is commonly involved in end-of-life care, when medical treatments and interventions no longer seem likely to benefit the patient, either by achieving longer survival or by enhancing their comfort and dignity. A few respondents were confident that available evidence can guide objective decision making in end-of-life situations, but most felt that the uncertainty that attends a patient’s terminal days encourages indecision about whether to regard a particular treatment as futile.
Futility is an absolute term; an intervention is either futile or it is not. If it is declared futile, a treatment should be suspended. This finality of the decision that a treatment is futile can disturb both doctor and patient. To the doctor it may bring a sense of failure and disappointment that they have nothing more to offer; the patient may experience despair and abandonment. Both have relied on treatments developed by advanced technologies that held great promise and built high expectations. Neither will wish to fracture a relationship established during the times when therapy achieved effective responses, and this may encourage the doctor to continue a treatment, even one now judged to be futile.
I suggest that, when assessing further management in such situations, “utility” is a more appropriate term than “futility”. Utility is not an absolute; it assesses usefulness over a range of applications and opportunities. An action may be partly useful, assisting one aspect of care but not another, or be temporarily useful, pending further consideration. It also has the advantage of positive intent, relating to what can or will be done rather than what should be avoided. In this sense it follows the suggestion made years ago to replace “not for resuscitation” notes with “good palliative care”.3
Utility is a concept that can attend all phases of patient progress. From the beginning it asks: “What useful outcome can I anticipate? What evidence can I marshal from the literature, from my own experience, and from my patient’s past responses and current hopes, to judge whether the action that I propose will be worthwhile?”
A consistent focus on utility has universal relevance to the conduct of any kind of therapy. It will seek to establish the point of balance between hoped for and undesired outcomes, between some good and no good at all. It will apply, for example, to the decision whether to prescribe antibiotics for a sore throat,4 or whether to request a prostate-specific antigen (PSA) test for a man with urogenital symptoms.5
That balance comes into an urgent focus when considering expensive and intrusive interventions in end-of-life situations. There, in particular, it must be weighed time and again, and shared with both the patient and their attending family.
It is the ethical duty of the physician to acknowledge the finality of “futile”, and to work around it by re-directing their efforts towards treatments that have utility. They will maximise the comfort and dignity of the patient and their grieving family, and may well bring an extension of days.6 To recognise and advise that there is always something that can be done will ease disappointment and maintain the therapeutic relationship. From 25 years of experience, I judge most palliative care to be just good medicine and within every doctor’s capability. It calls for kindness, attentiveness, comprehensive assessment of realities, awareness that specialist palliative care resources are available for difficult cases, and a personal confidence in the delivery of comfort care.7 It needs to be professed by the specialist in the hospital, the family physician serving the patient in the clinic, home or aged care facility, and by the many community care resources that families rely on. It represents a fundamental utility, and is everyone’s business.8
[Correspondence] Suicide rates in children and young people increase
The Office for National Statistics published the UK suicide rates for 2014 on Feb 4, 2016.1 Overall, 6122 people died by suicide in the UK; 120 (2%) fewer deaths than in 2013. For the first time, data on deaths for intentional self-harm in children aged 10–14 years have been included. The publication of these figures is welcomed and provides important information for public health and child and adolescent mental health services (CAMHS) about suicide rates in children and adolescents.2
[Correspondence] Questioning the outcome of the Building Blocks trial
Michael Robling and colleagues (Oct 13, p 146)1 are to be congratulated on conducting Building Blocks, a highly rigorous randomised controlled trial of the Family Nurse Partnership (FNP) programme in England, UK, and rightly highlighted the difficulty in showing changes similar to the US studies in a setting with comprehensive universal health services.1 The trial in the Netherlands,2 where FNP showed a positive effect on various primary outcomes, involved substantial adaptation of the programme to the local context, and was also more targeted in terms of risk.
Hospital cuts will hurt all
Households face a big jump in health costs and waiting times for treatment will blow out as a result of the Commonwealth’s cuts to public hospital funding.
A report on the impact of the Federal Government’s decision to slash public hospital spending by $57 billion from mid-2017 has found that households, state governments and private insurers will be forced to foot a growing share of the nation’s health bill, while public hospitals will have to increasingly ration their services, forcing many patients to seek treatment in the private system or face lengthy delays.
The analysis was undertaken by consultancy Ernst & Young at the behest of the South Australian Government, and focused on the effects of the slowdown in Commonwealth funding on the South Australian health system.
It found that spending on the state’s public hospitals was increasing at an average 6.7 per cent year, driven by the demands of an expanding but older and sicker population, as well as fixed costs like staff wages and improvements in technology.
But the Commonwealth’s revised funding formula, under which its expenditure will be indexed to population growth and inflation, means its contribution will grow at just 3.4 per cent a year.
If the State Government sustains its current rate of funding growth of 4.9 per cent a year, and the contribution from households and health funds continues to increase by 8.1 per cent a year, the Ernst & Young report warned this would leave a funding hole of 2 per cent a year.
It said this would force changes in the way public hospitals operate, with knock-on effects for the rest of the health system.
To cope with increasing financial constraints, hospitals will increasingly defer less critical or complex cases like tooth extractions and knee procedures, with an increasing proportion of their resources devoted to more complicated cases such as liver and heart transplants.
As a result, Ernst & Young estimated the number of separations handled would drop so that by 2019-20 more than 56,000 patients a year would be left untreated, reaching 107,000 a year by 2024-25.
Patients with less complex or serious ailments would face a choice of an increasingly long wait for treatment or, for those who could afford it, seeking care in the private system.
The report’s authors estimated that about one in five of those waiting for public hospital treatment would instead opt for the private sector, driving increased demand for private health insurance and adding 0.5 per cent a year to premiums.
This in turn would discourage younger, healthier people from taking out or maintaining private health cover, adding further upward pressure to premiums and increasing the cost for the Commonwealth of its private health insurance rebate scheme.
South Australian Premier Jay Weatherill said it was “an unavoidable fact” that the Commonwealth’s cuts would be felt hardest by the most vulnerable.
“But this is not just a South Australian problem,” he said. “This is something that affects every State in Australia. The states and territories simply cannot afford to bear the brunt of these cuts.”
See also: Hospital handout ‘dismally short’ of need
Adrian Rollins
In brief…
How to spend $20bn
Eminent medical researcher Professor Ian Frazer will lead a board charged with advising the Federal Government on investing funds from the $20 billion Medical Research Future Fund.
Professor Frazer, who will be joined by seven other directors drawn from the private sector and academia, will develop the five-year Australian Medical Research and Innovation Strategy, and set priorities every two years.
“The Advisory Board will ensure that any expenditure from the MRFF will have a strong business case, ensuring that the financial assistance provided…delivers the greatest value for all Australians,” Health Minister Sussan Ley said.
E-health overseer
The Commonwealth and the states and territories have agreed to set up the Australian Digital Health Agency to oversee the provision of national electronic health records and other digital health services.
The agency, which will begin operations in July, will be responsible for management of the national digital health strategy, and the design and operation of systems including the Commonwealth’s My Health Record.
Greens target ‘wasteful’ rebate
The Australian Greens would scrap the private health insurance rebate and reinvest the funds in public hospitals.
As political parties sharpen their policies ahead of the Federal election, the Greens have pledged to axe the “wasteful” PHI rebate, freeing up $10 billion over four years which would be redirected to the public hospital system.
Greens leader and public health specialist Dr Richard Di Natale said his party would also reinstate the joint Federal-State hospital funding model scrapped by the Coalition so that the Commonwealth would match 50 per cent of the efficient growth in hospital costs, with the change enshrined in law.
Trial run
The nation’s health ministers have committed to making Australia more attractive for clinical trials to boost investment and improve access to new medicines.
The ministers said that more needed to be done to make Australia a preferred location for clinical trials, including reducing fragmentation and inefficiencies. They have asked the Australian Health Ministers’ Advisory Council to develop options to organise sites, increase administrative efficiencies, improve engagement with sponsors, and reduce trial start-up times.
All the same
The nation’s hospitals will save $270 million over the next decade with the introduction of single standardised chart for the supply and reimbursement of Pharmaceutical Benefit Scheme medicines.
Commonwealth, state and territory health ministers have agreed to harmonise legislation to allow for the use of the standardised chart, in a move that will ease the regulatory burden on prescribers, pharmacists and nurses, improve patient safety and cut hospital administration overheads by around $27 a year. The new charts will be available from July.
A joint approach
The Commonwealth and the states will look at opportunities to jointly commission mental health services, including through Primary Healthcare Networks, following an agreement struck at the COAG Health Council meeting.
The agreement was suggested by the Queensland Government, which emphasised the need to identify opportunities for the joint commissioning of services across the Commonwealth and state-funded health services “[to] support a more integrated approach to service delivery and reduce any potential duplication”.
The meeting agreed that mental health was one of the areas where opportunities for joint commissioning would be explored, and called on PHNs to work with Local Health Networks to “align mental health commissioning efforts” from July.
Adrian Rollins
Assisted dying laws spread as euthanasia debate intensifies
Canada and California are set to join several European countries and US states in legalising doctor-assisted deaths amid calls for Australia to follow suit.
Canada is on track to allow physician-assisted dying from 6 June after the Trudeau Government introduced legislation to the Canadian Parliament, while the practice is set to become law in California on 9 June, nine months after a Bill was passed by the State’s legislature.
The international developments have come as debate about euthanasia in Australia intensifies.
High profile entertainer Andrew Denton has become a passionate advocate for legalising euthanasia, and last week he was joined by former Prime Minister Bob Hawke, who said it was “absurd” that patients in pain could not ask their doctor to help end their life.
“I think it is absurd that we should say that it is illegal that a person who is suffering terribly, and is in an irremediable condition, should be forced to continue to suffer,” Mr Hawke said in an interview on Mr Denton’s Better Off Dead podcast series. “It doesn’t meet any requirements of morality or good sense.”
Euthanasia will be debated in a policy session at the annual AMA National Conference next month, and the AMA’s Ethics and Medico-legal Committee is conducting a survey of member views on the issue as part of a review of the peak medical organisation’s policy on assisted dying.
Under the Canadian legislation, only adults with serious and irreversible medical conditions may seek a doctor-assisted death. They must apply in writing, with two witnesses, and the request must be evaluated by two doctors or nurses. Even once a request is granted there is a mandatory 15-day waiting period.
To prevent an influx of people from other countries seeking to avail themselves of the new law, it only applies to those eligible for Canadian Government-funded health services.
Under the new Californian laws, a person seeking assisted death must first have undergone rigorous questioning to determine that they were of sound mind and understood what they were seeking, and two doctors must have agreed that they had less than six months to live.
Opponents often fret that such laws will trigger a rash of doctor-assisted suicides, but experience in areas where they are in place suggests this is unlikely. In almost 20 years since similar legislation came into effect in Oregon, The Economist reported fewer than 1000 people have used it to take their own lives.
California’s Department of Health Care Services has estimated that in its first year, fewer than 450 seriously ill people will seek a prescription of lethal drugs through the Medicaid program, and even less will actually use them.
Adrian Rollins

 more_vert
more_vert